Interventional Solutions
That Make a Difference
Providing visionary medical devices to help caregivers deliver better patient outcomes.
Leading the Way
in Innovation
For more than 50 years, Argon medical devices has helped improve procedural efficiencies by developing innovative product lines for interventionalists and specialists across multiple fields. We’re relentlessly committed to improving efficacy for the caregivers we serve and the patients they treat.
FIND  SOLUTION
SOLUTION
 SOLUTION
SOLUTION
Featured Products
We innovate with a purpose: to create faster, smarter, easier and more effective ways to achieve your clinical goals and improve patient outcomes.


Argon Medical Opens New Asia-Pacific Operations Facility in Singapore

Argon Medical Streamlines Distribution of Product and Enhances Customer Training for Europe, Asia, and Africa with the Debut of a Modern, Mixed-Use Facility

Argon Medical Expands Oncology Product Offering with Acquisition of the SeQure® and DraKon™ Microcatheters
Argon Medical Announces First Patient Enrollment in CLEAN-PE, a Prospective, Multicenter Study of a New Treatment for Pulmonary Embolism
Argon Medical Announces the Expansion of Liver Management Portfolio with Two New Product Introductions

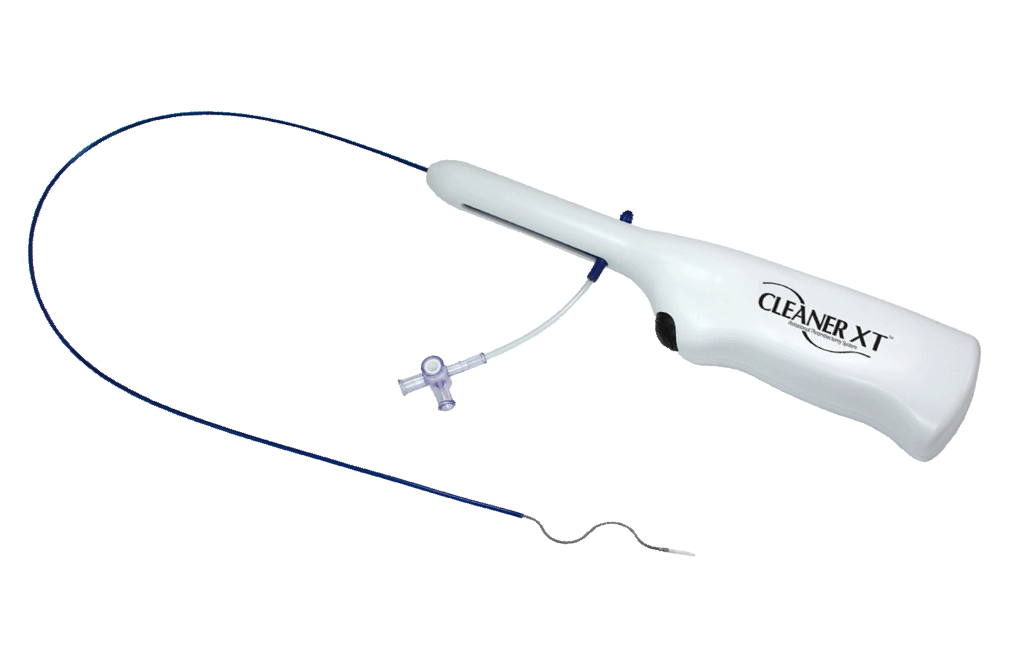
Argon Medical Launches CLEANER Vac™ Thrombectomy System to Remove Blood Clot in the Peripheral Venous Vasculature